Bohr’s Solution for Hydrogen
Bohr was able to derive the formula for the hydrogen spectrum using basic physics, the planetary model of the atom, and some very important new proposals. His first proposal is that only certain orbits are allowed: We say that the orbits of electrons in atoms are quantized. Each orbit has a different energy, and electrons can move to a higher orbit by absorbing energy and drop to a lower orbit by emitting energy. If the orbits are quantized, the amount of energy absorbed or emitted is also quantized, producing discrete spectra. Photon absorption and emission are among the primary methods of transferring energy into and out of atoms. The energies of the photons are quantized, and their energy is explained as being equal to the change in energy of the electron when it moves from one orbit to another. In equation form, this is
13.19 Here, is the change in energy between the initial and final orbits, and is the energy of the absorbed or emitted photon. It is quite logical, that is, expected from our everyday experience, that energy is involved in changing orbits. A blast of energy is required for the space shuttle, for example, to climb to a higher orbit. What is not expected is that atomic orbits should be quantized. This is not observed for satellites or planets, which can have any orbit given the proper energy (see Figure 13.17).
Figure 13.18 shows an energy-level diagram, a convenient way to display energy states. In the present discussion, we take these to be the allowed energy levels of the electron. Energy is plotted vertically with the lowest or ground state at the bottom and with excited states above. Given the energies of the lines in an atomic spectrum, it is possible, although sometimes very difficult, to determine the energy levels of an atom. Energy-level diagrams are used for many systems, including molecules and nuclei. A theory of the atom or any other system must predict its energies based on the physics of the system.
Bohr was clever enough to find a way to calculate the electron orbital energies in hydrogen. This was an important first step that has been improved upon, but it is well worth repeating here, because it does correctly describe many characteristics of hydrogen. Assuming circular orbits, Bohr proposed that the angular momentum of an electron in its orbit is quantized, that is, it has only specific, discrete values. The value for is given by the formula
13.20
where is the angular momentum,
is the electron’s mass,
is the radius of the
th orbit, and
is Planck’s constant. Note that angular momentum is
For a small object at a radius
and
so that
Quantization says that this value of
can only be equal to etc. At the time, Bohr himself did not know why angular momentum should be quantized, but using this assumption he was able to calculate the energies in the hydrogen spectrum, something no one else had done at the time.
From Bohr’s assumptions, we will now derive a number of important properties of the hydrogen atom from the classical physics we have covered in the text. We start by noting the centripetal force causing the electron to follow a circular path is supplied by the Coulomb force. To be more general, we note that this analysis is valid for any single-electron atom. So, if a nucleus has protons for hydrogen, 2 for helium, etc.) and only one electron, that atom is called a hydrogen-like atom. The spectra of hydrogen-like ions are similar to hydrogen, but shifted to higher energy by the greater attractive force between the electron and nucleus. The magnitude of the centripetal force is while the Coulomb force is The tacit assumption here is that the nucleus is more massive than the stationary electron, and the electron orbits about it. This is consistent with the planetary model of the atom. Equating these
13.21 Angular momentum quantization is stated in an earlier equation. We solve that equation for substitute it into the above, and rearrange the expression to obtain the radius of the orbit. This yields
13.22 where is defined to be the Bohr radius, since for the lowest orbit and for hydrogen It is left for this chapter’s Problems and Exercises to show that the Bohr radius is
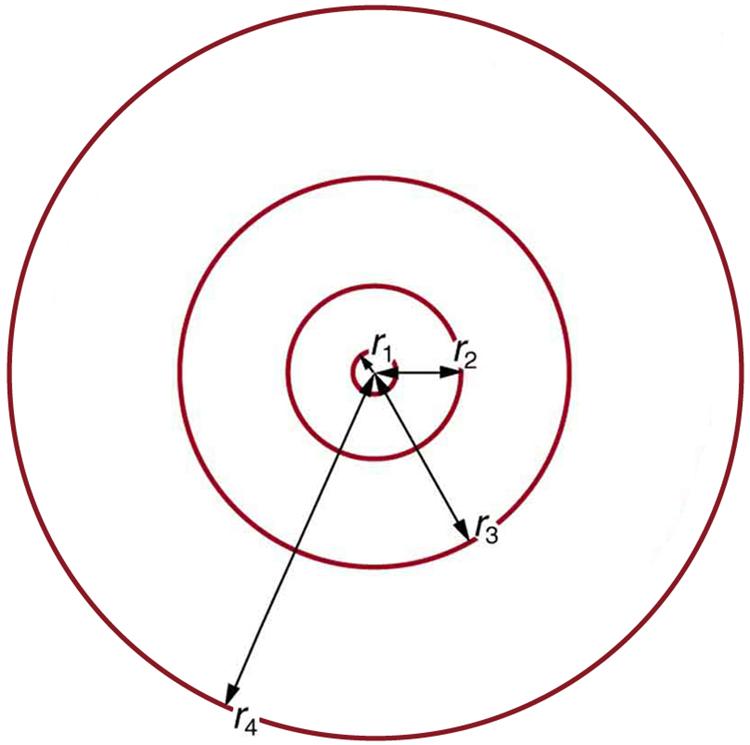
13.23 These last two equations can be used to calculate the radii of the allowed (quantized) electron orbits in any hydrogen-like atom. It is impressive that the formula gives the correct size of hydrogen, which is measured experimentally to be very close to the Bohr radius. The earlier equation also tells us that the orbital radius is proportional to as illustrated in Figure 13.19.
To get the electron orbital energies, we start by noting that the electron energy is the sum of its kinetic and potential energy
13.24 Kinetic energy is the familiar assuming the electron is not moving at relativistic speeds. Potential energy for the electron is electrical, or where is the potential due to the nucleus, which looks like a point charge. The nucleus has a positive charge ; thus,
recalling an earlier equation for the potential due to a point charge. Since the electron’s charge is negative, we see that Entering the expressions for and we find
13.25 Now we substitute and from earlier equations into the above expression for energy. Algebraic manipulation yields
13.26
for the orbital energies of hydrogen-like atoms. Here, is the ground-state energy for hydrogen and is given by
13.27 Thus, for hydrogen,
13.28 Figure 13.20 shows an energy-level diagram for hydrogen that also illustrates how the various spectral series for hydrogen are related to transitions between energy levels.
Electron total energies are negative, since the electron is bound to the nucleus, analogous to being in a hole without enough kinetic energy to escape. As approaches infinity, the total energy becomes zero. This corresponds to a free electron with no kinetic energy, since gets very large for large and the electric potential energy thus becomes zero. Thus, 13.6 eV is needed to ionize hydrogen to go from –13.6 eV to 0, or unbound, an experimentally verified number. Given more energy, the electron becomes unbound with some kinetic energy. For example, giving 15.0 eV to an electron in the ground state of hydrogen strips it from the atom and leaves it with 1.4 eV of kinetic energy.
Finally, let us consider the energy of a photon emitted in a downward transition, given by the equation to be
13.29 Substituting we see that
13.30 Dividing both sides of this equation by gives an expression for
13.31 It can be shown that
13.32 is the Rydberg constant. Thus, we have used Bohr’s assumptions to derive the formula first proposed by Balmer years earlier as a recipe to fit experimental data.
13.33
We see that Bohr’s theory of the hydrogen atom answers the question as to why this previously known formula describes the hydrogen spectrum. It is because the energy levels are proportional to where is a non-negative integer. A downward transition releases energy, and so must be greater than The various series are those where the transitions end on a certain level. For the Lyman series, —that is, all the transitions end in the ground state (see also Figure 13.20). For the Balmer series, or all the transitions end in the first excited state and so on. What was once a recipe is now based in physics, and something new is emerging—angular momentum is quantized.