The Electron
Gas discharge tubes, such as that shown in Figure 13.4, consist of an evacuated glass tube containing two metal electrodes and a rarefied gas. When a high voltage is applied to the electrodes, the gas glows. These tubes were the precursors to today’s neon lights. They were first studied seriously by Heinrich Geissler, a German inventor and glassblower, starting in the 1860s. The English scientist William Crookes, among others, continued to study what for some time were called Crookes tubes, wherein electrons are freed from atoms and molecules in the rarefied gas inside the tube and are accelerated from the cathode—negative—to the anode—positive—by the high potential. These cathode rays collide with the gas atoms and molecules and excite them, resulting in the emission of EM radiation that makes the electrons’ path visible as a ray that spreads and fades as it moves away from the cathode.
Gas discharge tubes today are most commonly called cathode ray tubes, because the rays originate at the cathode. Crookes showed that the electrons carry momentum—they can make a small paddle wheel rotate. He also found that their normally straight path is bent by a magnet in the direction expected for a negative charge moving away from the cathode. These were the first direct indications of electrons and their charge.
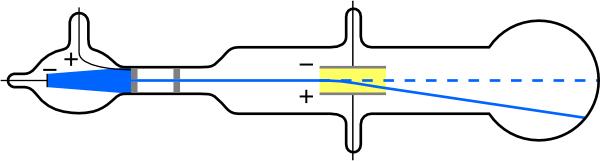
The English physicist J. J. Thomson (1856–1940) improved and expanded the scope of experiments with gas discharge tubes (see Figure 13.5 and Figure 13.6). He verified the negative charge of the cathode rays with both magnetic and electric fields. Additionally, he collected the rays in a metal cup and found an excess of negative charge. Thomson was also able to measure the ratio of the charge of the electron to its mass, —an important step to finding the actual values of both and Figure 13.7 shows a cathode ray tube, which produces a narrow beam of electrons that passes through charging plates connected to a high-voltage power supply. An electric field is produced between the charging plates, and the cathode ray tube is placed between the poles of a magnet so that the electric field is perpendicular to the magnetic field of the magnet. These fields, being perpendicular to each other, produce opposing forces on the electrons. As discussed for mass spectrometers in More Applications of Magnetism, if the net force due to the fields vanishes, then the velocity of the charged particle is In this manner, Thomson determined the velocity of the electrons and then moved the beam up and down by adjusting the electric field.
To see how the amount of deflection is used to calculate note that the deflection is proportional to the electric force on the electron.
13.1 But the vertical deflection is also related to the electron’s mass, since the electron’s acceleration is
13.2
The value of is not known, since was not yet known. Substituting the expression for electric force into the expression for acceleration yields
13.3
Gathering terms, we have
13.4
The deflection is analyzed to get and is determined from the applied voltage and distance between the plates; thus, can be determined. With the velocity known, another measurement of can be obtained by bending the beam of electrons with the magnetic field. Since we have Consistent results are obtained using magnetic deflection.
What is so important about the ratio of the electron’s charge to its mass? The value obtained is
13.5 This is a huge number, as Thomson realized, and it implies that the electron has a very small mass. It was known from electroplating that about is needed to plate a material, a factor of about 1,000 less than the charge per kilogram of electrons. Thomson went on to do the same experiment for positively charged hydrogen ions—now known to be bare protons—and found a charge per kilogram about 1,000 times smaller than that for the electron, implying that the proton is about 1,000 times more massive than the electron. Today, we know more precisely that
13.6 where is the charge of the proton and is its mass. This ratio—to four significant figures—is 1,836 times less charge per kilogram than for the electron. Since the charges of electrons and protons are equal in magnitude, this implies
Thomson performed a variety of experiments using differing gases in discharge tubes and employing other methods, such as the photoelectric effect, for freeing electrons from atoms. He always found the same properties for the electron, proving it to be an independent particle. For his work, the important pieces of which he began to publish in 1897, Thomson was awarded the 1906 Nobel Prize in Physics. In retrospect, it is difficult to appreciate how astonishing it was to find that the atom has a substructure. Thomson himself said, “It was only when I was convinced that the experiment left no escape from it that I published my belief in the existence of bodies smaller than atoms.”
Thomson attempted to measure the charge of individual electrons, but his method could determine its charge only to the order of magnitude expected.
Since Faraday’s experiments with electroplating in the 1830s, it had been known that about 100,000 C per mole was needed to plate singly ionized ions. Dividing this by the number of ions per mole, that is, by Avogadro’s number, which was approximately known, the charge per ion was calculated to be about close to the actual value.
An American physicist, Robert Millikan (1868–1953) (see Figure 13.8) decided to improve upon Thomson’s experiment for measuring and was eventually forced to try another approach, which is now a classic experiment performed by students. The Millikan oil drop experiment is shown in Figure 13.9.
In the Millikan oil drop experiment, fine drops of oil are sprayed from an atomizer. Some of these are charged by the process and can then be suspended between metal plates by a voltage between the plates. In this situation, the weight of the drop is balanced by the electric force
13.7
The electric field is produced by the applied voltage, hence, and is adjusted to just balance the drop’s weight. The drops can be seen as points of reflected light using a microscope, but they are too small to directly measure their size and mass. The mass of the drop is determined by observing how fast it falls when the voltage is turned off. Since air resistance is very significant for these submicroscopic drops, the more massive drops fall faster than the less massive, and sophisticated sedimentation calculations can reveal their mass. Oil is used rather than water, because it does not readily evaporate, and so mass is nearly constant. Once the mass of the drop is known, the charge of the electron is given by rearranging the previous equation
13.8
where is the separation of the plates and is the voltage that holds the drop motionless. The same drop can be observed for several hours to see that it really is motionless. By 1913, Millikan had measured the charge of the electron to an accuracy of 1 percent, and he improved this by a factor of 10 within a few years to a value of He also observed that all charges were multiples of the basic electron charge and that sudden changes could occur in which electrons were added or removed from the drops. For this very fundamental direct measurement of and for his studies of the photoelectric effect, Millikan was awarded the 1923 Nobel Prize in Physics.
With the charge of the electron known and the charge-to-mass ratio known, the electron’s mass can be calculated. It is
13.9 Substituting known values yields
13.10 or
13.11 where the round-off errors have been corrected. The mass of the electron has been verified in many subsequent experiments and is now known to an accuracy of better than one part in one million. It is an incredibly small mass and remains the smallest known mass of any particle that has mass. (Some particles, such as photons, are massless and cannot be brought to rest, but travel at the speed of light.) A similar calculation gives the masses of other particles, including the proton. To three digits, the mass of the proton is now known to be
13.12 which is nearly identical to the mass of a hydrogen atom. What Thomson and Millikan had done was to prove the existence of one substructure of atoms, the electron, and further to show that it had only a tiny fraction of the mass of an atom. The nucleus of an atom contains most of its mass, and the nature of the nucleus was completely unanticipated.
Another important characteristic of quantum mechanics was also beginning to emerge. All electrons are identical to one another. The charge and mass of electrons are not average values; rather, they are unique values that all electrons have. This is true of other fundamental entities at the submicroscopic level. All protons are identical to one another, and so on.